The Tracker™ introduces a next-generation everting balloon system built for smooth, guided advancement.
The Tracker™ introduces a next-generation everting balloon system built for smooth, guided advancement.
A first-in-class balloon design engineered to navigate naturally through
the Eustachian tube with a minimally invasive approach.
Watch how the everting balloon mechanism advances and adapts
throughout deployment.

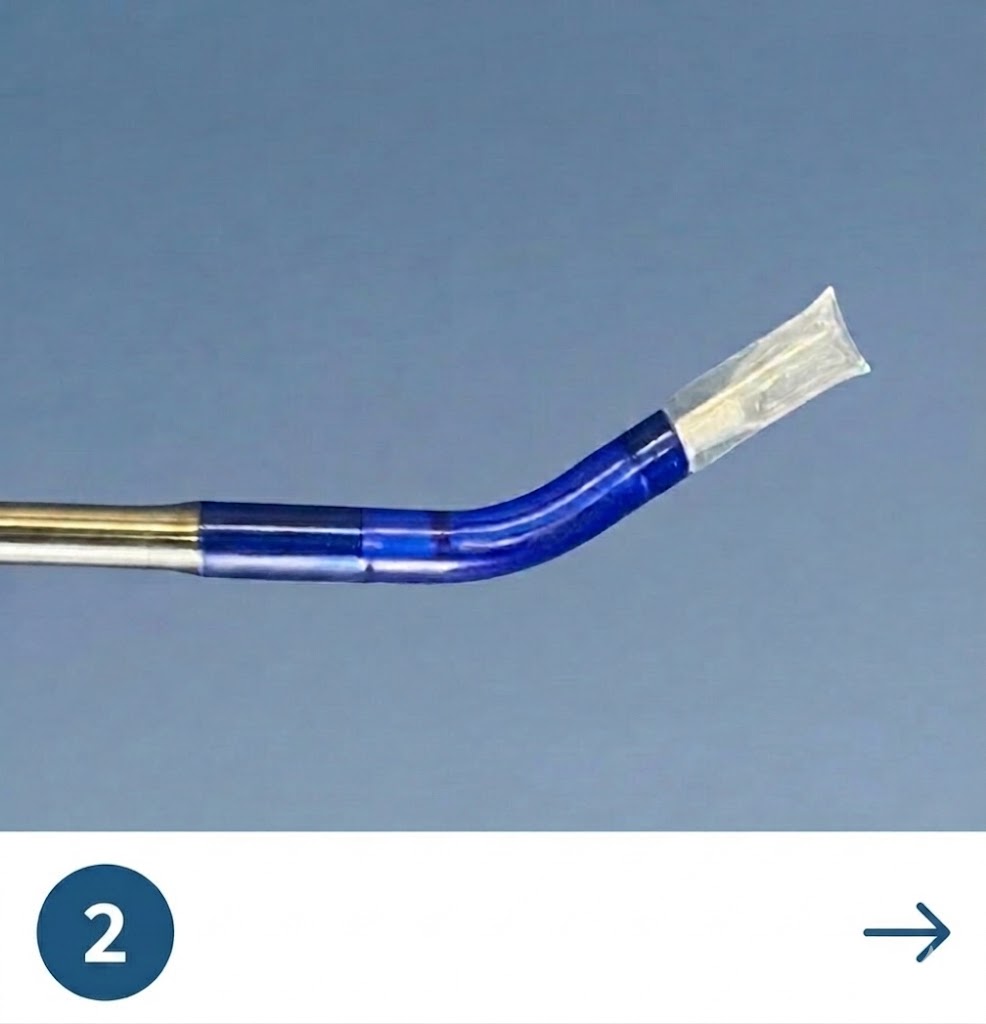
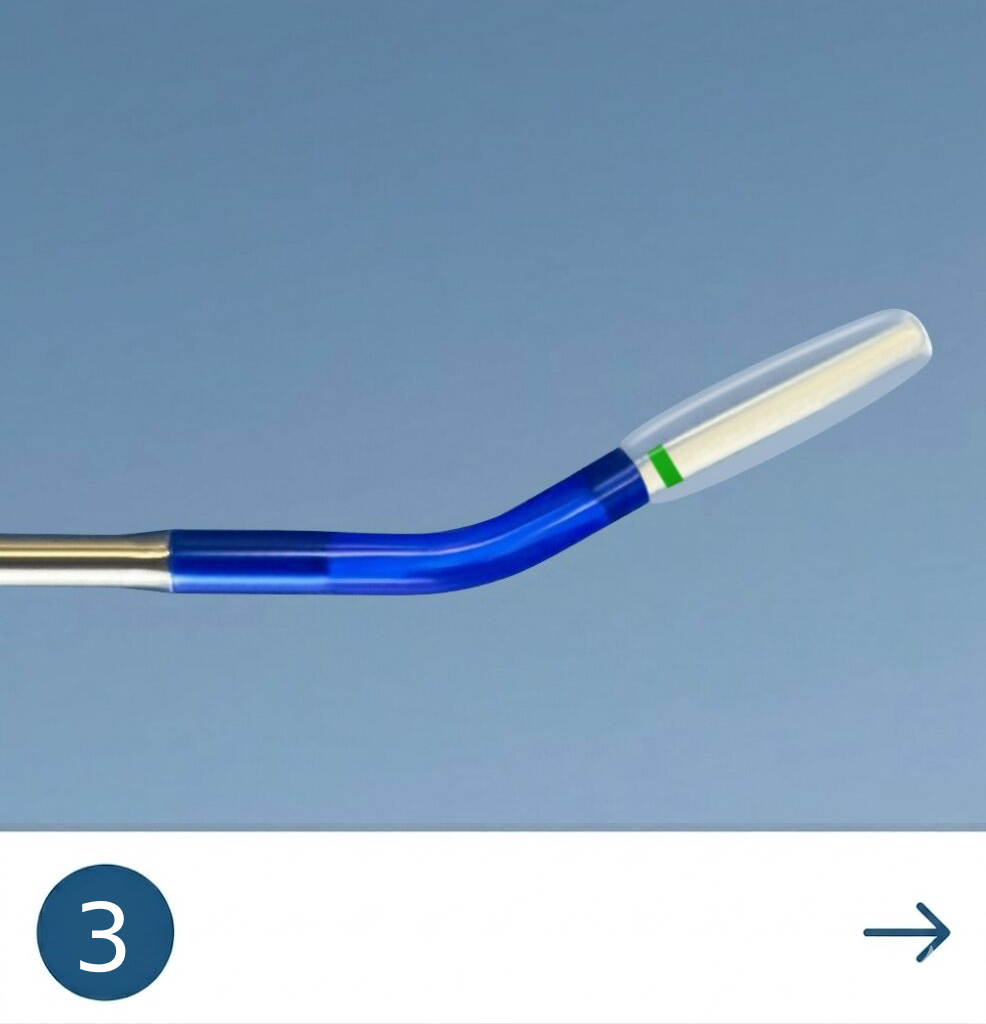




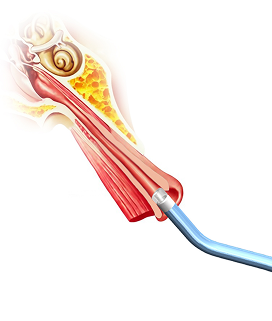
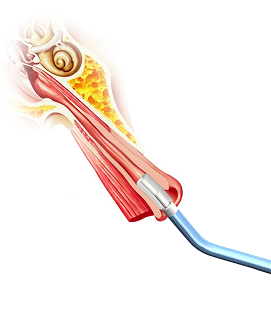
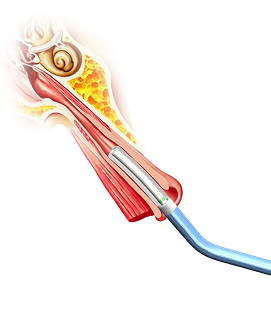
The Tracker™ advances with a self-navigating everting balloon mechanism
designed to conform naturally during placement.

A balloon-only approach
designed without any stiff or
rigid components inside the
Eustachian tube

Engineered to naturally advance
through anatomy while reducing
dependence on pre-placed
guidance systems.

An innovative everting
mechanism designed to support
controlled advancement and
placement.

Designed to address
Eustachian tube dysfunction
with a streamlined balloon-only
system.
Receive launch updates, development news, and future
announcements from Dalent Medical.


